The Science Behind Project Hail Mary’s Infrared Detection Is More Real Than You’d Expect
In This Article
What Project Hail Mary Gets Right About Infrared (And Why It Matters More Than You Think)
The film’s central detection mechanism isn’t just good science fiction. It’s a working principle that engineers use every day to catch invisible threats before they become catastrophic ones.
Okay, so you just watched Project Hail Mary and now you have questions about infrared. Good. That means the movie did its job.
Whether you walked out of the theater genuinely curious about the science or you’re a thermal imaging professional who spent two hours alternating between nodding and wanting to fact-check the screen, this piece is for you. We’re going to work through the real physics behind what the film depicts, where it earns its credibility, where it takes creative license, and what the infrared principles at the core of the story actually look like when applied to problems here on Earth.
Fair warning: this is going to get technical. But in a good way. The kind of technical where things start to make satisfying sense.
First: Is the Petrova Line Actually Plausible?
The Petrova line is the infrared signature stretching between the Sun and Venus that tips off scientists to the existence of Astrophage. It is fictional. But the underlying concept, that a biological or chemical entity could have a specific, detectable infrared absorption or emission signature that makes it visible in one part of the spectrum while remaining invisible in others, is entirely grounded in real science. Andy Weir was precise about this: in the story, Astrophage emits infrared energy at 25.984 micrometers, a very specific far-infrared wavelength, and maintains a constant temperature of 96.415 degrees Celsius as it converts stellar energy into mass. The instrument Ryland Grace uses to detect this signature is called the Petrovascope, a specialized sensor tuned specifically to that wavelength. None of that is arbitrary worldbuilding. It reflects how real spectral detection actually works.
Every molecule interacts with infrared radiation in a characteristic way. This is the basis of infrared spectroscopy, one of the most widely used analytical techniques in chemistry and materials science. Molecules absorb infrared energy at specific wavelengths that correspond to their molecular bond vibrations. Carbon dioxide absorbs strongly at 4.26 micrometers. Methane has a signature near 3.3 micrometers. Water vapor absorbs across several bands. Each molecule has what amounts to an infrared fingerprint.
So the idea that Astrophage would have a specific wavelength at which it absorbs and emits infrared energy, making it detectable against the background of space at that wavelength and essentially transparent at others, is scientifically coherent. We actually do discover gases in planetary atmospheres, interstellar clouds, and astronomical objects by looking for exactly these kinds of spectral signatures. The detection of water vapor on exoplanets, the identification of organic molecules in nebulae, the analysis of Martian atmospheric composition, all of it works on this principle.
The Petrova line is not real. The detection method that would find it, if it were, absolutely is.
Why Can Infrared Detect Things That Visible Light Cannot?
Here is the thing about visible light: it is a tiny sliver of reality.
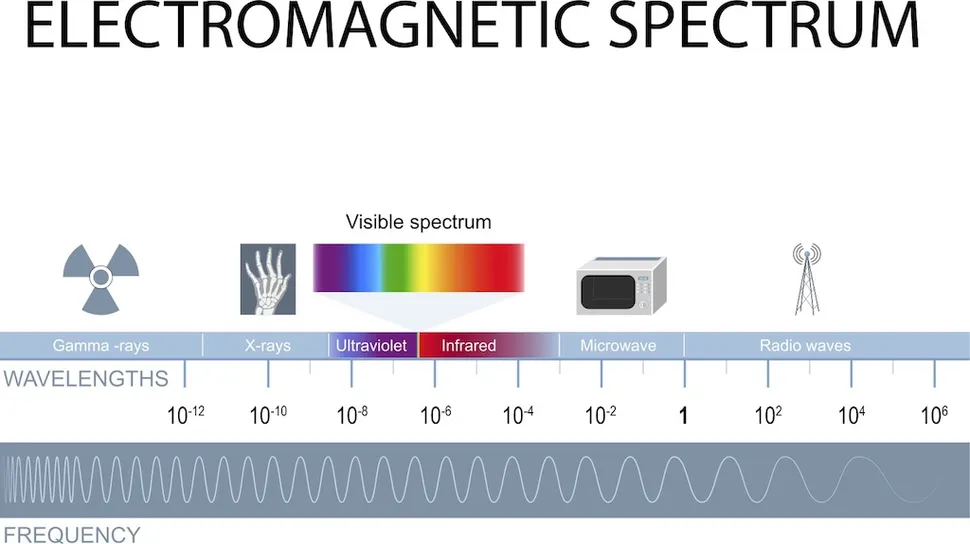
The electromagnetic spectrum spans an enormous range of wavelengths and frequencies, from radio waves with wavelengths measured in kilometers down to gamma rays with wavelengths smaller than an atom. The human eye is sensitive to a band between roughly 380 and 700 nanometers. That is it. Everything outside of that window, which is most of the electromagnetic spectrum, is invisible to us without instruments.
Credit: Live Science
Credit: Live Science
Infrared radiation occupies the range from about 700 nanometers to 1 millimeter, sitting just beyond the red end of the visible spectrum. What makes it particularly useful for detection is a physical law called Planck’s law, which describes how objects emit radiation as a function of their temperature. Every object above absolute zero emits thermal radiation. The warmer the object, the more energy it emits and the shorter the peak wavelength of that emission.
At the surface temperature of the Sun, peak emission falls in the visible range, which is why we can see sunlight. At room temperature, say 20 degrees Celsius, an object’s peak emission is around 10 micrometers, deep in the thermal infrared. To a thermal camera, a person standing in a dark room is glowing. A machine running hot stands out sharply against its surroundings. A battery cell beginning to overheat looks completely different from one operating normally, even if they appear identical to the eye.
This is the core capability: infrared detection reveals thermal contrast. Where visible light shows you surface reflectance, texture, and color, infrared shows you temperature distribution. Those are different kinds of information, and in many situations, especially situations involving safety and equipment health, temperature is the more important signal.
Why Do Objects Emit Infrared at All?
If you are going to understand thermal imaging properly, you need two concepts: blackbody radiation and emissivity. They sound intimidating. They are not.
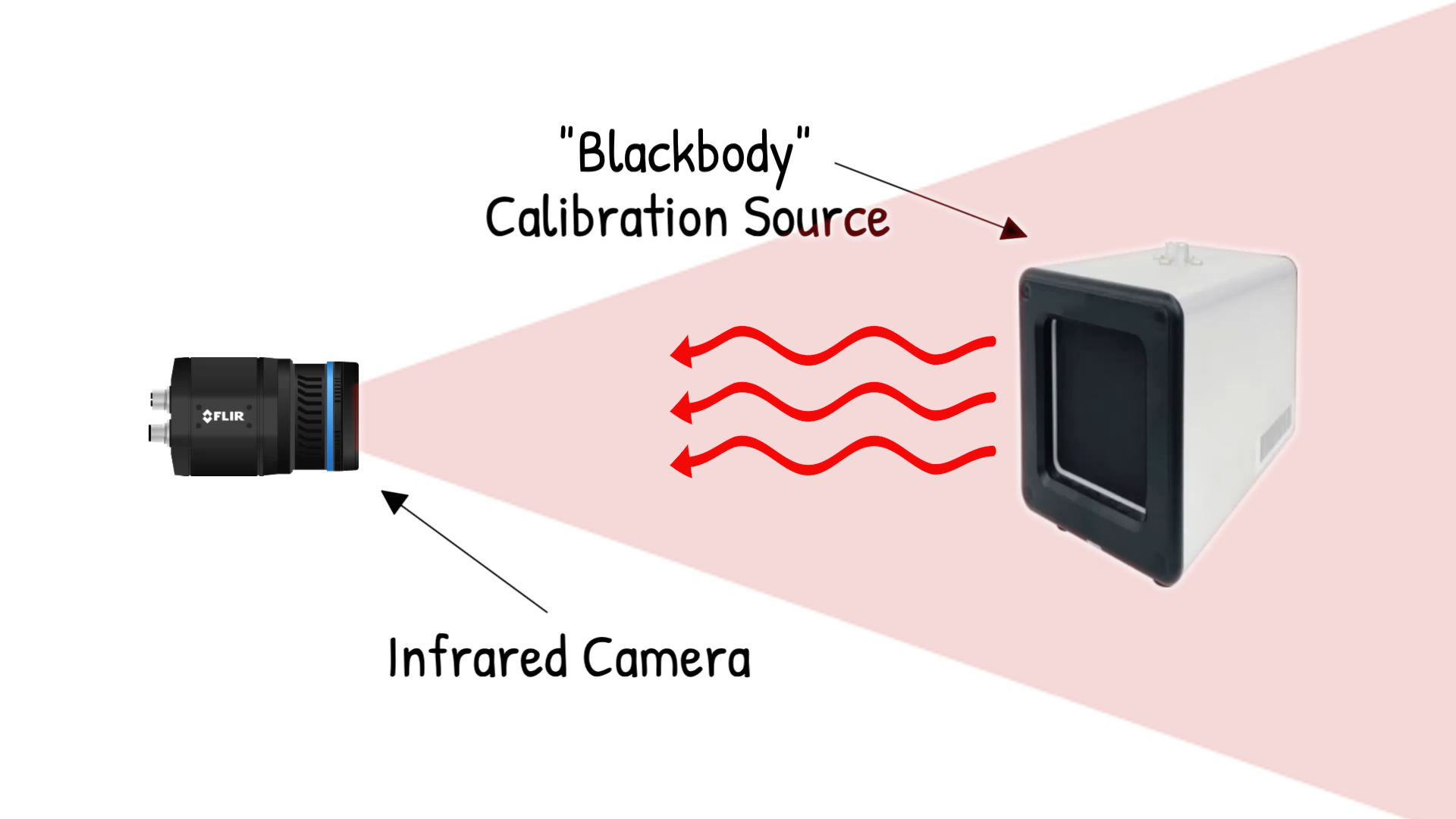
A blackbody is a theoretical object that absorbs all incoming radiation and emits the maximum possible thermal energy for its temperature. It is a perfect emitter and a perfect absorber. No real object is a true blackbody, but the concept gives physicists a clean baseline to work from. Planck’s law describes exactly how much energy a blackbody radiates at each wavelength for a given temperature, and it is the equation behind every thermal camera calibration in the world.
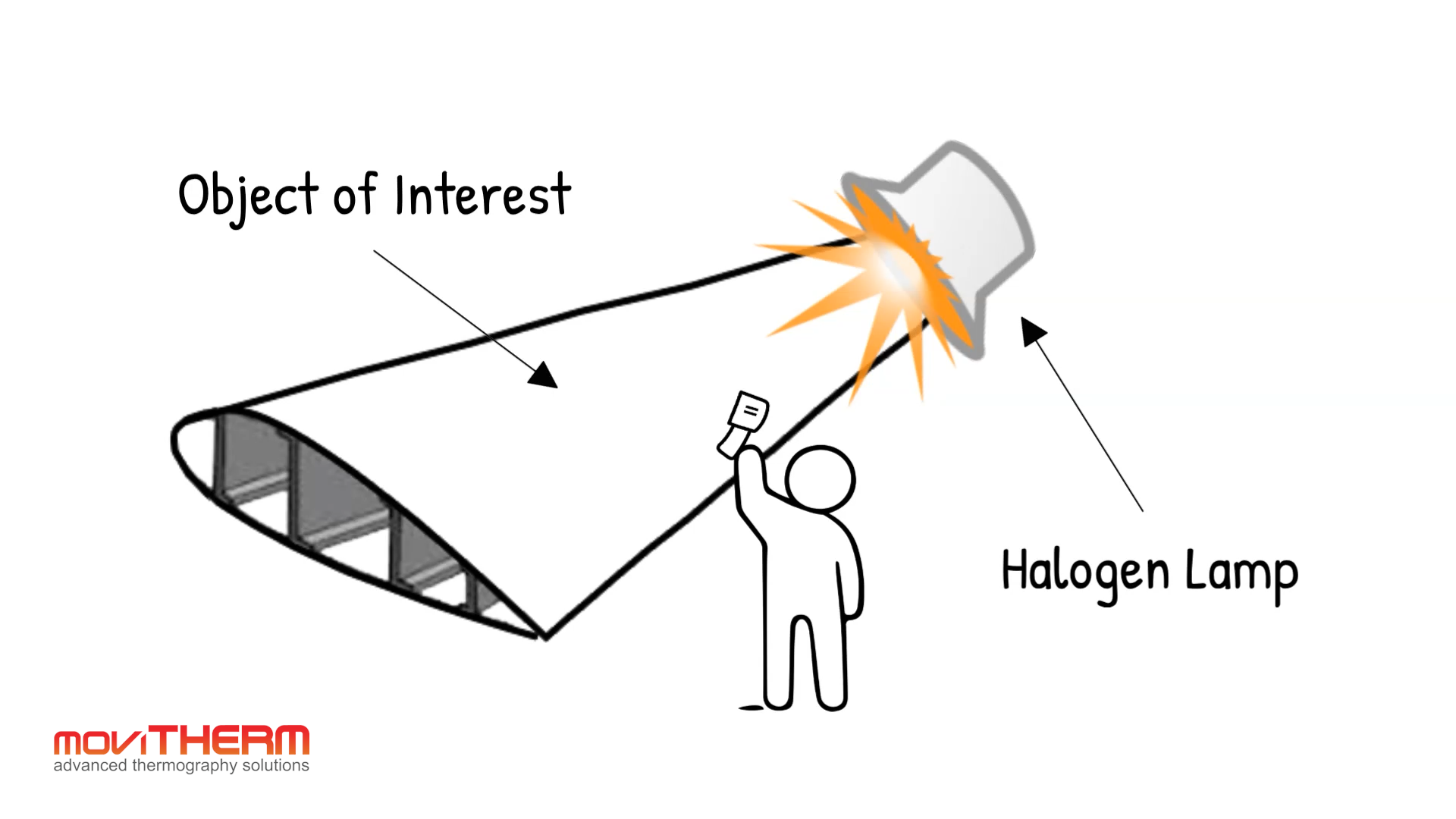
Emissivity is where things get interesting for practical thermal imaging. Emissivity is a number between 0 and 1 that describes how efficiently a real surface emits thermal radiation compared to a perfect blackbody at the same temperature. A blackbody has an emissivity of 1.0. Human skin is around 0.98, which is why thermal cameras read body temperature with high accuracy. Painted metal surfaces typically fall between 0.85 and 0.95. Bare, polished metals are a different story — a polished aluminum surface can have an emissivity as low as 0.05, meaning it emits only 5 percent of the thermal energy a blackbody would at the same temperature.
This matters enormously in the field. A thermal camera pointed at a polished metal surface is not giving you an accurate temperature reading unless emissivity is correctly accounted for. Highly reflective surfaces also pick up thermal radiation from surrounding objects and reflect it back at the camera, which can produce completely misleading readings. Experienced thermographers know to look for these conditions and either correct for them or reposition the camera. It is one of the reasons thermal imaging is a discipline, not just a tool.
In the film’s context, Astrophage presumably has a consistent emissivity at its detection wavelength, which is part of what makes the Petrova line identifiable as a distinct signature rather than noise. The organism emits predictably. Real-world thermal targets behave the same way — predictable emissivity is what makes repeatable, reliable thermal measurement possible.
The Infrared Spectrum Isn’t One Thing. It’s Several.
One of the things the film handles well, at least implicitly, is that detecting Astrophage requires a specific wavelength. Not just “infrared” broadly, but a particular band within it. That specificity matters, because different parts of the infrared spectrum behave differently and are suited to different detection tasks.
Near-Infrared (NIR): 0.75 – 1.4 micrometers
NIR sits just past the edge of what the human eye can see and is used extensively in machine vision, product sorting, agricultural sensing, and spectroscopic analysis. NIR cameras can detect differences in material composition and moisture content that are invisible in visible light. Silicon-based sensors work well here, which makes NIR cameras relatively affordable and widely deployed.
Short-Wave Infrared (SWIR): 1.4 – 3 micrometers
SWIR is valuable for applications requiring penetration through atmospheric haze or moisture. It is used in outdoor inspection, semiconductor wafer analysis, solar cell inspection, and some food quality applications. Here is the part that genuinely surprises people: certain plastics that look completely solid in visible light are effectively transparent in SWIR. Once you know that, it changes how you think about inspection entirely. You are not just seeing differently — you are seeing through things that looked closed.
Mid-Wave Infrared (MWIR): 3 – 8 micrometers
MWIR detects high-temperature objects and hot gases with exceptional sensitivity. Jet engine inspection, flame detection in petrochemical facilities, and gas leak detection all fall naturally into the MWIR range. If you need to detect a hydrogen flame, which is nearly invisible to the naked eye, a MWIR camera will find it immediately.
Long-Wave Infrared (LWIR): 8 – 14 micrometers
LWIR is the workhorse of thermal imaging for ambient temperature monitoring. At practical operating temperatures, objects emit their peak thermal radiation in this band — but the 8 to 14 micrometer range is not defined by emission physics alone. It also aligns with a key atmospheric transmission window, meaning the atmosphere absorbs relatively little energy here, which is what makes LWIR cameras practical for real-world use at distance. This combination of peak emission and atmospheric transparency is why LWIR sensors dominate electrical inspection, building diagnostics, predictive maintenance, condition monitoring of industrial equipment, and early fire detection. Most fixed thermal monitoring systems in industrial environments use LWIR for exactly this reason.
Choosing the right band is not a minor technical detail. It is the difference between detecting the phenomenon you care about and producing an image that tells you nothing. The scientists in the film tune their instruments to the Petrova wavelength. Real thermal engineers make the same kind of decision every time they specify a camera for an application. And here is a detail worth noting: Astrophage’s emission wavelength of roughly 26 micrometers falls outside the standard LWIR band of 8 to 14 micrometers entirely. It sits in the far infrared, a range that conventional thermal cameras are not designed to detect. This is precisely why the Petrovascope had to be a specialized instrument. A standard thermal camera would not have found it. That is not a plot convenience, it is an accurate reflection of why band selection and instrument specification matter so much in real detection work.
Could We Actually Detect Something Like Astrophage in Real Life?
Let’s take the premise seriously for a moment, because it deserves it.
Astrophage is a fictional microorganism that consumes energy from stars and emits it at a specific infrared wavelength. The detection challenge in the film is identifying a faint, consistent infrared signature against the overwhelming background radiation of the Sun. This is actually a well-understood problem in observational astronomy called signal-to-noise ratio, and it is the same challenge faced in spectroscopy, remote sensing, and atmospheric science.
Space telescopes including the James Webb Space Telescope do exactly what the scientists in the film are doing. They analyze infrared spectral signatures from distant objects to determine atmospheric and surface composition, identify molecules, and detect anomalies against background radiation. JWST operates primarily in the near- and mid-infrared bands and has already identified specific molecular signatures in exoplanet atmospheres at distances measured in light-years.
If Astrophage were real and had a distinct infrared spectral signature, the detection method depicted in the film would be scientifically appropriate. The hard part would not be the detection principle. It would be the sensitivity required to distinguish that signature from noise at that distance, and the computational work to confirm that the signal is real, consistent, and anomalous relative to known physics. These are the same problems astronomers and spectroscopists work on today.
The film compresses this process considerably. Real spectral anomaly confirmation would take longer than a single observation. But the underlying science is not wrong.
And here is the connection that tends to land quietly but stick: the signal-to-noise challenge Petrova faced, distinguishing a real anomaly from background radiation and confirming it is consistent over time rather than a one-off artifact, is the same fundamental problem that industrial thermal monitoring solves on a much smaller scale every day. A thermal camera watching a battery rack at a transit depot is doing the same thing JWST does when it stares at an exoplanet atmosphere. It is looking for a signature that should not be there, confirming it is real, and triggering a response before the situation changes in ways that cannot be undone.
What Infrared Detection Looks Like Here on Earth
The film makes infrared detection feel like a cosmic-scale problem. In practice, the same principles play out at the scale of a factory floor, a transit depot, a recycling facility, or an electrical substation — and the stakes, while less cinematic, are very real.
Early Fire Detection
Fire does not begin as flame. It begins as heat. In industrial environments like recycling facilities, waste management operations, and bulk material handling, the heat build-up stage can begin hours before any visible smoke or flame appears. A fixed LWIR thermal camera monitoring a conveyor belt or storage bunker continuously will detect that temperature rise in its earliest stage, before a smoke detector has anything to respond to. This is the detection window that matters most, and it is only accessible through thermal imaging. moviTHERM’s iTL platform is built specifically around this principle, combining fixed thermal cameras with automated alarm logic and remote monitoring to maintain 24/7 visibility into thermal conditions without requiring someone to be on-site watching a screen.
Battery and Thermal Runaway Monitoring
Lithium-ion battery systems, in electric transit vehicles, battery energy storage installations, and EV charging infrastructure, present one of the most time-sensitive thermal detection challenges in modern industry. Thermal runaway, the self-reinforcing exothermic reaction that can escalate from a localized hotspot to an uncontrollable fire in seconds, begins with a temperature anomaly that is detectable in the infrared well before any other indicator appears. The parallel to Astrophage’s 96.415-degree constant temperature is almost uncomfortably direct: the threat has a thermal signature, it is consistent, and the only way to catch it early is to be looking at the right wavelength continuously. moviTHERM works with energy storage facilities to deploy thermal monitoring systems that do exactly this.
Predictive Maintenance and Condition Monitoring
Electric motors, transformers, switchgear, bearings, and conveyor drives all exhibit characteristic thermal signatures when moving toward failure. A motor running with bearing wear heats differently than a healthy one. A transformer with a failing connection shows elevated temperature at the fault point. None of this is visible to the eye, but to a thermal camera calibrated to the right emissivity and mounted at the right angle, the signature is clear and measurable over time. moviTHERM works with operations teams and OEMs to integrate thermal sensing into maintenance workflows before failures occur rather than after.
Non-Destructive Testing (NDT)
Infrared thermography is also a primary tool in non-destructive testing for composite materials, aerospace components, and structural inspection. When a composite panel is stressed or delaminated internally, it conducts heat differently from an intact panel. Active thermography, applying a controlled heat source and imaging how the surface temperature evolves over time, can reveal subsurface defects with no contact and no damage to the part. This is the same spectral logic the film uses: the anomaly is invisible until you look at the right wavelength under the right conditions.
Across all of these applications, the underlying discipline is the same as what Petrova and Grace were practicing: select the right band, account for emissivity, distinguish signal from noise, and build the response infrastructure around what the instrument tells you. The scale is different. The physics is identical.
The Film Got It Right Where It Mattered
Project Hail Mary is not a documentary. Astrophage is not real, the Petrova line does not exist, and no one is currently launching a lone scientist toward Tau Ceti on a desperate species-saving mission. We would like to be clear about that.
But the film’s central premise, that infrared detection is the tool that reveals what nothing else can, that the earliest and most critical signal of a dangerous process in motion is a thermal signature, and that catching it requires both the right instrument and the right analytical infrastructure to interpret what that instrument is seeing, is not science fiction at all.
It is thermal engineering. It is spectroscopy. It is the reason fixed thermal cameras exist on conveyor lines, in battery storage facilities, and above recycling bunkers.
Ryland Grace figured out that you have to look in the right part of the spectrum. That is, genuinely, the whole job.

About moviTHERM
Founded in 1999 and headquartered in Irvine, California, moviTHERM is a leading provider of thermal imaging and remote monitoring solutions for early fire detection, condition monitoring, non-destructive testing, and quality inspection. The company designs and deploys complete thermal monitoring systems — integrating best-in-class infrared hardware with its own software platforms to serve clients across industrial, aerospace, energy, and manufacturing markets.